HOW to make a Blood Smear:
Whether for examination in-clinic, for submission to the lab to interpret alongside in-clinic haematology data, or to accompany an EDTA sample to the lab for full CBC, a good, well-spread smear is required to allow for more accurate estimations, observations, and differentials.
The BLOOD
- The EDTA blood should be fresh and at room temperature.
- Invert the tube gently several times to mix before making the smear.
The SLIDES
- Good quality spreader slides are vital and should have a cut, not ground, glass edge – run your fingernail along the edge of the slide, it should feel smooth not rough. No matter how perfect a technique, a good smear cannot be made with a rough-edged spreader slide!
- The quality of the slide on which the smear is made is less important.
The SMEAR
- Place a drop (~5 mm diameter) of well mixed blood at one end of the slide. DO NOT allow the drop to dry out!
- Place the spreader slide on the sample slide in front of the blood drop and draw it back into the blood.
- Wait for the blood to extend under the spreader slide to within 1 mm of the edges of the slide, then push forward smoothly and quickly.

- Do not place too much pressure on the spreader or many of the WBCs will end up in the feathered edge.
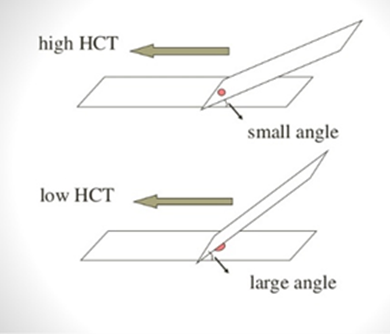
- Angle the spreader slide according to the haematocrit – with most bloods, a 45° angle is fine but for thinner blood (e.g., anaemic animals) use a larger angle, and for thicker blood (e.g., dehydrated animals) a smaller angle.
- The smear should cover ½ to ¾ of the slide.
- Aim for a rounded distal edge.
- For smears that are to be submitted to the lab, ensure the feathered edge of the blood smear does not extend to within ~15 mm of the end of the slide, as this area is not stained by the automatic stainer.
- Allow to air dry thoroughly.
- Do not place a wet smear into a slide container, as the blood may shift on the slide and cause red blood cells (RBCs) to clump together in certain areas, creating a false appearance of rouleaux formation. Red cells may also lyse.
- Do not store your smear in the fridge as the cells will be affected by condensation.
WHY make a blood smear?
For in-clinic analysis – how often do you look at a blood smear or send it to the lab for evaluation when putting a sample through your in-clinic haematology analyser? Every sample? Or just when something odd shows up? A study in 2013 (Cora et. al., 2013) found 20% of smears detected abnormalities missed by the analyser. This percentage increases if the scatter diagram is not interpreted in conjunction with the numerical results. Modern analysers are machines working from computer-based algorithms and as such work from a limited range of data points. Accuracy is greatest when patient parameters are normal, and falls dramatically in samples with uncommon abnormalities.
If you are not sending smears to your lab, it is even more important to look at every smear. How do you learn to recognise “abnormal” if “normal” is not soundly understood?
For laboratory analysis – when sending an EDTA sample to a commercial lab for a CBC it may seem redundant to make a blood smear as well. After all, the staff at the lab are experienced at making smears, so why go to the trouble when an expert will have the blood within the next 12 to 24 hours?
As EDTA has no preservative and is hypertonic, it is damaging to cells. Red blood cell (RBC) morphology is stable for about 6 hours (likely shorter in hot weather), after which crenation, sphering, and fragmentation begin. This can make it difficult to detect ecchinocytes, acanthocytes, Heinz bodies, spherocytes, and RBC parasites. White blood cells (WBC) begin to degenerate within 3 hours and show severe degeneration by 12 to 18 hours. Degenerative changes seen in aged WBCs can mimic those found in an inflammatory leukogram, with cytoplasmic vacuolation and nuclear swelling occurring. More severe cellular degeneration may eventually occur, rendering cells unidentifiable.
Refrigeration of whole blood (EDTA) sample slows the deterioration of cell morphology (but note, smears should NOT be refrigerated).
Reference: MC Cora, JA Neel, CB Grindem, GE Kissling, PR Hess. Comparison of automated versus manual neutrophil counts for the detection of cellular abnormalities in dogs receiving chemotherapy: 50 cases (May to June 2008). J Am Vet Med Assoc. 242:1539-43, 2013
Reviewed: April 2024
